CHM 130 Introduction to Chemistry at Arizona College of Nursing
Access The Exact Questions for CHM 130 Introduction to Chemistry at Arizona College of Nursing
💯 100% Pass Rate guaranteed
🗓️ Unlock for 1 Month
Rated 4.8/5 from over 1000+ reviews
- Unlimited Exact Practice Test Questions
- Trusted By 200 Million Students and Professors
What’s Included:
- Unlock Actual Exam Questions and Answers for CHM 130 Introduction to Chemistry at Arizona College of Nursing on monthly basis
- Well-structured questions covering all topics, accompanied by organized images.
- Learn from mistakes with detailed answer explanations.
- Easy To understand explanations for all students.

Free CHM 130 Introduction to Chemistry at Arizona College of Nursing Questions
Comparing the solubilities of the two compounds 1-hexanamine and heptane. 1-hexanamine is ______
-
not soluble in water, while heptane is
-
less soluble in water than heptane
-
equally soluble in water as heptane
-
more soluble in water than heptane
Explanation
Correct Answer: D. more soluble in water than heptane
Explanation of Correct Answer:
1-hexanamine is more soluble in water than heptane because it contains an –NH₂ (amine) group capable of forming hydrogen bonds with water molecules. This polar interaction enhances its solubility. Heptane, on the other hand, is a nonpolar hydrocarbon lacking any functional groups that can interact with water, making it highly hydrophobic and virtually insoluble. Therefore, the polar nature of 1-hexanamine explains its greater solubility in water.
Which of the following is a disaccharide?
-
Glucose
-
Galactose
-
Fructose
-
Maltose
Explanation
Correct Answer: D. Maltose
Explanation of Correct Answer:
Maltose is a disaccharide composed of two glucose molecules joined by a glycosidic bond. Disaccharides form when two monosaccharides undergo a dehydration (condensation) reaction, releasing water. Maltose is commonly produced during the breakdown of starch in digestion or fermentation. In contrast, glucose, galactose, and fructose are single-unit sugars (monosaccharides) that cannot be hydrolyzed into simpler sugars.
What is a characteristic of alkanes?
-
Each carbon atom is bonded to four other atoms, and all bonds are single bonds.
-
Carbon atoms form a ring structure with alternating single and double bonds.
-
They contain at least one carbon-to-carbon triple bond.
-
They contain at least one carbon-to-carbon double bond.
Explanation
Correct Answer: A. Each carbon atom is bonded to four other atoms, and all bonds are single bonds.
Explanation of Correct Answer:
Alkanes are saturated hydrocarbons, meaning all carbon-carbon bonds are single covalent bonds (σ-bonds). Each carbon atom forms four single bonds with hydrogen or other carbon atoms, giving them a tetrahedral geometry. Alkenes (option D) contain double bonds, and alkynes (option C) contain triple bonds. Option B describes aromatic hydrocarbons, not alkanes.
Using IUPAC nomenclature, identify this compound:
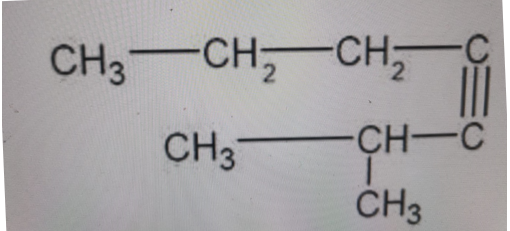
-
6-methyl-4-heptyne
-
2-methyl-3-heptyne
-
2,2-diethyl-4-heptyne
-
1-isopropyl-4-butyne
Explanation
Correct Answer: B. 2-methyl-3-heptyne
Explanation of Correct Answer:
The longest continuous chain has seven carbons, so the parent name is heptyne. The triple bond is internal, located between carbons 3 and 4 when numbered to give the lowest possible locant for the triple bond, so it is hept-3-yne (written 3-heptyne). There is a methyl (CH₃) substituent on carbon 2, giving 2-methyl-3-heptyne. The other names either place the methyl incorrectly, use the wrong parent chain, or use non-IUPAC substituent names.
Name the following functional group presented in this compound.
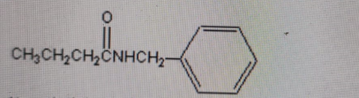
-
Amine
-
Amide
-
Alcohol
-
Ester
Explanation
Correct Answer: B. Amide
Explanation of Correct Answer:
The structure shows a carbonyl group (C=O) directly attached to a nitrogen (–C(=O)–NH–). This is the defining feature of an amide functional group. An amine would have a nitrogen attached to carbon without a carbonyl. An alcohol would contain an –OH group, which is not present here. An ester would have –C(=O)–O–R, with an oxygen bonded to the carbonyl carbon, which this compound does not have.
Determine the IUPAC name of this structure:
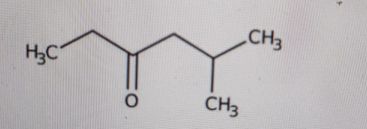
-
3-pentanone-5-methyl
-
5-methyl-3-hexanone
-
2-methyl-4-hexanone
-
5-methyl-pentanal
Explanation
Correct Answer: B. 5-methyl-3-hexanone
Explanation of Correct Answer:
First, identify the longest continuous carbon chain that includes the carbonyl carbon. The chain has six carbons, so the parent name is hexanone. The carbonyl (C=O) is in the middle of the chain, on carbon 3, so that gives us 3-hexanone. Next, look at the right side of the chain: that carbon has an extra CH₃ branch. That branch is on carbon 5 of the main chain. A methyl group on carbon 5 gives 5-methyl. Put it all together: 5-methyl-3-hexanone.
Which structure below is trans-2-butene?
-
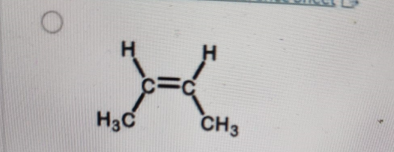
-
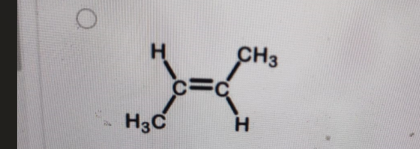
-
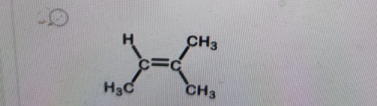
-
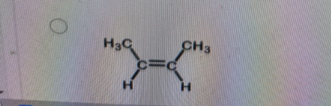
Explanation
Correct Answer: B. (second structure)
Explanation of Correct Answer:
In trans-2-butene, the two methyl (CH₃) groups attached to the double-bonded carbons are on opposite sides of the double bond. This spatial arrangement reduces steric repulsion and is characteristic of the trans isomer. In contrast, cis-2-butene has both CH₃ groups on the same side of the double bond. The second structure clearly shows the CH₃ groups opposite each other—confirming it as trans-2-butene.
Which of the following is the major disaccharide that occurs naturally in milk and milk products?
-
Lactose
-
Sucrose
-
Maltose
-
Cellobiose
Explanation
Correct Answer: A. Lactose
Explanation of Correct Answer:
Lactose is the primary disaccharide found naturally in milk and milk products. It is composed of one glucose molecule and one galactose molecule joined by a β(1→4) glycosidic bond. During digestion, the enzyme lactase breaks down lactose into these monosaccharides for absorption. Individuals who lack sufficient lactase experience lactose intolerance. Other disaccharides—such as sucrose (in plants), maltose (in starch breakdown), and cellobiose (in cellulose)—are not predominant in milk.
What is the correct classification for the alcohol shown below?
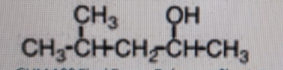
-
Secondary
-
Primary
-
Quaternary
-
Tertiary
Explanation
Correct Answer: A. Secondary
Explanation of Correct Answer:
The hydroxyl (–OH) group is attached to a carbon that is itself attached to two other carbons. The structure is CH₃–CH(CH₃)–CH₂–CH(OH)–CH₃. The carbon bearing the –OH is bonded to the CH₂ group on the left and a CH₃ group on the right, so it has two carbon neighbors. That makes it a secondary alcohol. A primary alcohol would have the –OH on a carbon attached to only one other carbon, and a tertiary alcohol would have it on a carbon attached to three other carbons. "Quaternary" is not used for alcohol classification.
A substance that functions to bind to an enzyme's active site and decrease its catalytic activity is known as a(an):
-
Inhibitor
-
Substrate
-
Affector
-
Activator
Explanation
Correct Answer: A. Inhibitor
Explanation of Correct Answer:
An inhibitor is a molecule that binds to an enzyme and reduces its catalytic activity. Competitive inhibitors specifically bind to the enzyme’s active site, preventing the substrate from binding. This decreases the rate of product formation. Inhibitors are important in regulating metabolic pathways and can be reversible or irreversible, depending on the type of interaction between the enzyme and inhibitor.
How to Order
Select Your Exam
Click on your desired exam to open its dedicated page with resources like practice questions, flashcards, and study guides.Choose what to focus on, Your selected exam is saved for quick access Once you log in.
Subscribe
Hit the Subscribe button on the platform. With your subscription, you will enjoy unlimited access to all practice questions and resources for a full 1-month period. After the month has elapsed, you can choose to resubscribe to continue benefiting from our comprehensive exam preparation tools and resources.
Pay and unlock the practice Questions
Once your payment is processed, you’ll immediately unlock access to all practice questions tailored to your selected exam for 1 month .