CHEM 120 W8 Exam3 and W8 Lab Practicum at Chamberlain University
Access The Exact Questions for CHEM 120 W8 Exam3 and W8 Lab Practicum at Chamberlain University
💯 100% Pass Rate guaranteed
🗓️ Unlock for 1 Month
Rated 4.8/5 from over 1000+ reviews
- Unlimited Exact Practice Test Questions
- Trusted By 200 Million Students and Professors
What’s Included:
- Unlock Actual Exam Questions and Answers for CHEM 120 W8 Exam3 and W8 Lab Practicum at Chamberlain University on monthly basis
- Well-structured questions covering all topics, accompanied by organized images.
- Learn from mistakes with detailed answer explanations.
- Easy To understand explanations for all students.

Free CHEM 120 W8 Exam3 and W8 Lab Practicum at Chamberlain University Questions
Which of the following best describes the terms "independent variable" and "dependent variable"?
-
The independent variable is the cause, and the dependent variable is the effect
-
The dependent variable is the cause, and the independent variable is the effect.
-
The independent variable is held constant as a positive control, and the dependent variable is the negative control.
-
There is no relationship between the independent variable and the dependent variable.
Explanation
The independent variable is the one that the researcher manipulates or changes to observe its effect on another variable. The dependent variable is the measurable outcome that responds to these changes. In other words, the independent variable causes the dependent variable to vary. For example, in testing plant growth, the amount of sunlight (independent) affects the height of the plant (dependent).
Select all of the following that you would expect to be a liquid oil at room temperature:
-
A fatty acid containing multiple carbon-to-carbon double bonds
-
A dipeptide
-
A fatty acid containing hexane
-
A fatty acid classified as unsaturated
-
A carbohydrate with a glycosidic bond
Explanation
A. A fatty acid containing multiple carbon-to-carbon double bonds
Fatty acids with multiple double bonds (polyunsaturated fatty acids) have kinks in their hydrocarbon chains that prevent tight packing of molecules. This results in weaker intermolecular forces, keeping them liquid at room temperature. Examples include omega-3 and omega-6 fatty acids found in vegetable oils and fish oils.
D. A fatty acid classified as unsaturated
Unsaturated fatty acids (both mono- and polyunsaturated) contain one or more double bonds, which reduce molecular rigidity and melting point. Thus, they are typically liquid oils at room temperature, unlike saturated fats, which are solid
The condensed structural formula for the compound ethene is:
-
CH₂CH₂
-
CH₄
-
CCl₄
-
CH₃CH₃
Explanation
Ethene (also called ethylene) is a hydrocarbon with the formula C₂H₄. It contains a double bond between the two carbon atoms, making it an alkene. The condensed structural formula is written as CH₂=CH₂ or CH₂CH₂ (with the understanding that a double bond is present).
How do we convert from moles (mol) of a compound to mass (g) of a compound? I'll give you a hint: You're going to need the molar weight (MW).
-
g = mol × MW
-
g = mol ÷ MW
-
MW = g × mol
-
g = MW ÷ mol
Explanation
To find the mass of a compound in grams, multiply the number of moles by the compound’s molar mass (MW), which represents grams per mole. This relationship links the amount of substance to its measurable weight:
mass (g) = moles × molar mass (g/mol).
For example, 2 moles of water (H₂O) with a molar mass of 18.0 g/mol would have a mass of 36.0 g.
Which of the following functional groups is used as an important component of hand sanitizers?
-
Carboxylic acid
-
Alcohol
-
Alkane
-
Ketone
Explanation
Alcohols, particularly ethanol and isopropanol, are the primary active ingredients in most hand sanitizers. They work by denaturing proteins and dissolving lipids in microbial cell membranes, effectively killing bacteria and viruses. Concentrations of 60–70% alcohol are proven to be most effective for disinfection while remaining safe for skin use.
Blood is known to be a buffered solution in the body. Does the intake of acidic or alkaline foods significantly affect the blood pH?
-
No, the blood pH is constant.
-
Yes, the pH increases after eating.
-
Yes, acidic food decreases pH and alkaline food increases it.
-
No, the blood pH fluctuates randomly.
Explanation
The human body maintains blood pH within a narrow range of about 7.35–7.45 through buffer systems such as the bicarbonate–carbonic acid system. These buffers neutralize excess acids or bases to prevent significant pH changes. While food may temporarily alter the pH of saliva or urine, blood pH remains tightly regulated by the lungs and kidneys, ensuring stable physiological conditions necessary for enzyme function and cellular processes.
Which of the compounds below is classified as a carboxylic acid?
-
CH₃CH₂COOH
-
CH₃CH₂CH₂OH
-
CH₃CH₂OCH₃
-
CH₃COOCH₂CH₃
Explanation
A carboxylic acid is an organic compound containing the carboxyl functional group (-COOH). The formula CH₃CH₂COOH represents propanoic acid, which includes this group. Carboxylic acids are known for their acidic properties and often have a sour taste and sharp odor.
Why does the correct Lewis structure of CO2 involve a double bond between each of the oxygen atoms and the carbon atom?
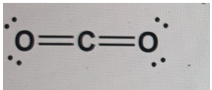
-
To complete the octet for carbon
-
Because the oxygens would otherwise have more than eight electrons
-
Because carbon is more electronegative than oxygen
-
To emphasize that it's a covalent bond
Explanation
In the Lewis structure of CO₂, carbon needs four additional electrons to complete its octet, while each oxygen needs two. By forming two double bonds—one between carbon and each oxygen—both the carbon and oxygen atoms achieve stable octets. This arrangement minimizes formal charges and accurately represents the molecule’s bonding, showing that CO₂ is a linear molecule with strong covalent bonds between atoms.
The enzyme OREOase acts on chocolate sandwich cookies to break the cookies apart to get to the creamy center. Occasionally, a vanilla cookie also fits into the OREOase active site, but the enzyme cannot act on the vanilla cookie so the reaction halts. Eventually, the vanilla cookie leaves the active site. What is the best description of the vanilla cookie in this scenario?
-
Competitive inhibitor
-
Denaturing of the protein
-
Cofactor
-
Coenzyme
-
Non-competitive inhibitor
Explanation
A competitive inhibitor is a substance that resembles the enzyme’s normal substrate and competes for binding at the active site. In this case, the vanilla cookie fits into OREOase’s active site just like the chocolate cookie (the true substrate) but does not undergo a reaction. This temporarily blocks the enzyme’s activity until the inhibitor leaves, allowing the correct substrate to bind again.
Which is the best description of rRNA?
-
It is part of the ribosome
-
It holds the entire genetic code
-
It carries a copy of the DNA code to the ribosome
-
It carries the amino acid to the ribosome
Explanation
Ribosomal RNA (rRNA) forms the structural and functional core of ribosomes, the cellular organelles responsible for protein synthesis. It helps align messenger RNA (mRNA) and transfer RNA (tRNA) during translation and catalyzes the formation of peptide bonds between amino acids. rRNA thus plays a direct enzymatic and structural role in building proteins, making it essential for gene expression and cellular function.
How to Order
Select Your Exam
Click on your desired exam to open its dedicated page with resources like practice questions, flashcards, and study guides.Choose what to focus on, Your selected exam is saved for quick access Once you log in.
Subscribe
Hit the Subscribe button on the platform. With your subscription, you will enjoy unlimited access to all practice questions and resources for a full 1-month period. After the month has elapsed, you can choose to resubscribe to continue benefiting from our comprehensive exam preparation tools and resources.
Pay and unlock the practice Questions
Once your payment is processed, you’ll immediately unlock access to all practice questions tailored to your selected exam for 1 month .